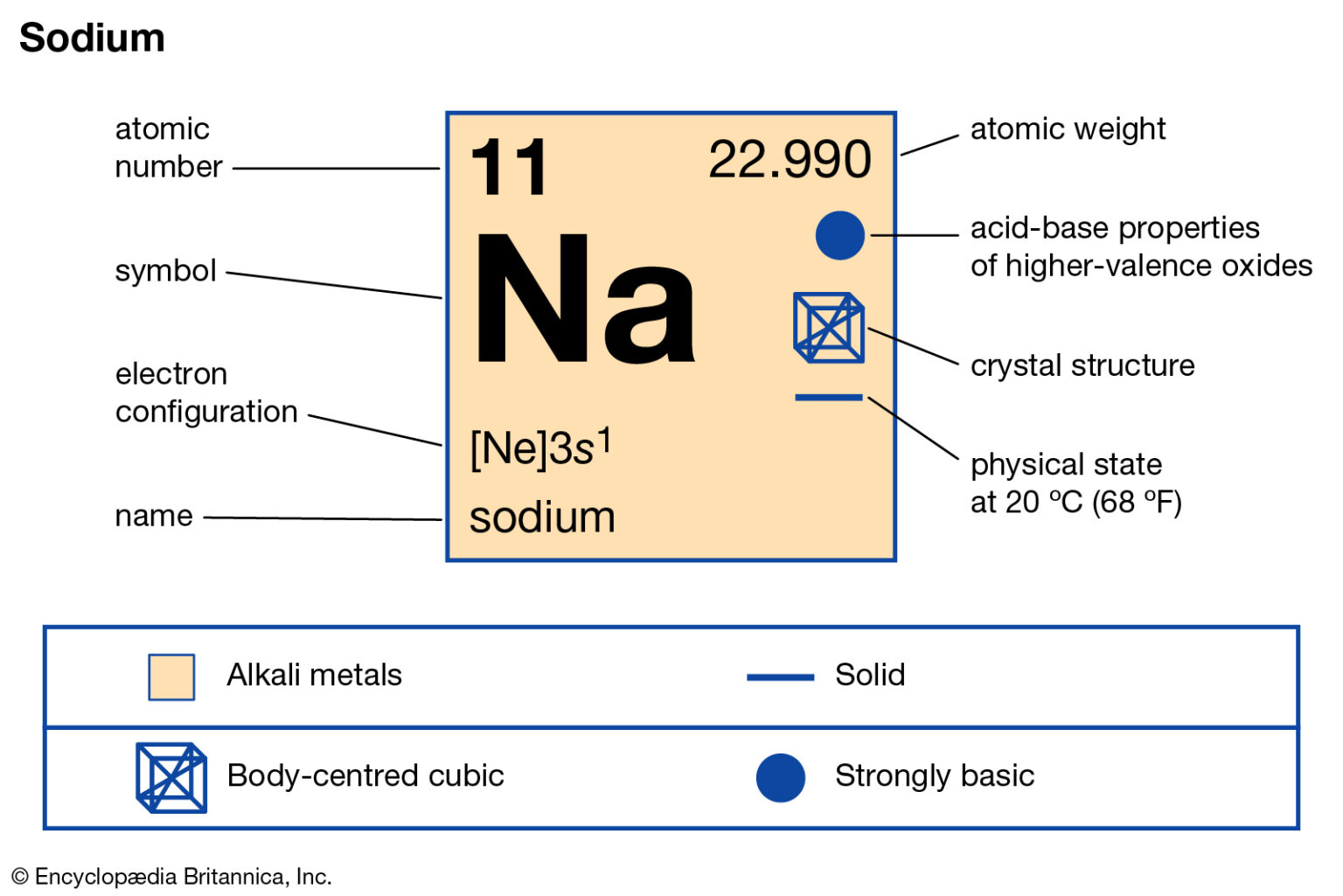
The third shell has three subshells, labeled s, p, and d. Because the second shell has only two subshells, atoms with more electrons now must begin the third shell. With neon, the 2 p subshell is completely filled. Thus, the electron configurations for the next six atoms are as follows: Now that the 2 s subshell is filled, electrons in larger atoms start filling the 2 p subshell. The next largest atom, beryllium, has 4 electrons, so its electron configuration is 1 s 22 s 2. Figure 2.6.1 Shell diagram of lithium (Li) atom. The shell closest to the nucleus (first shell) has 2 dots representing the 2 electrons in 1 s, while the outermost shell ( 2 s) has 1 electron. 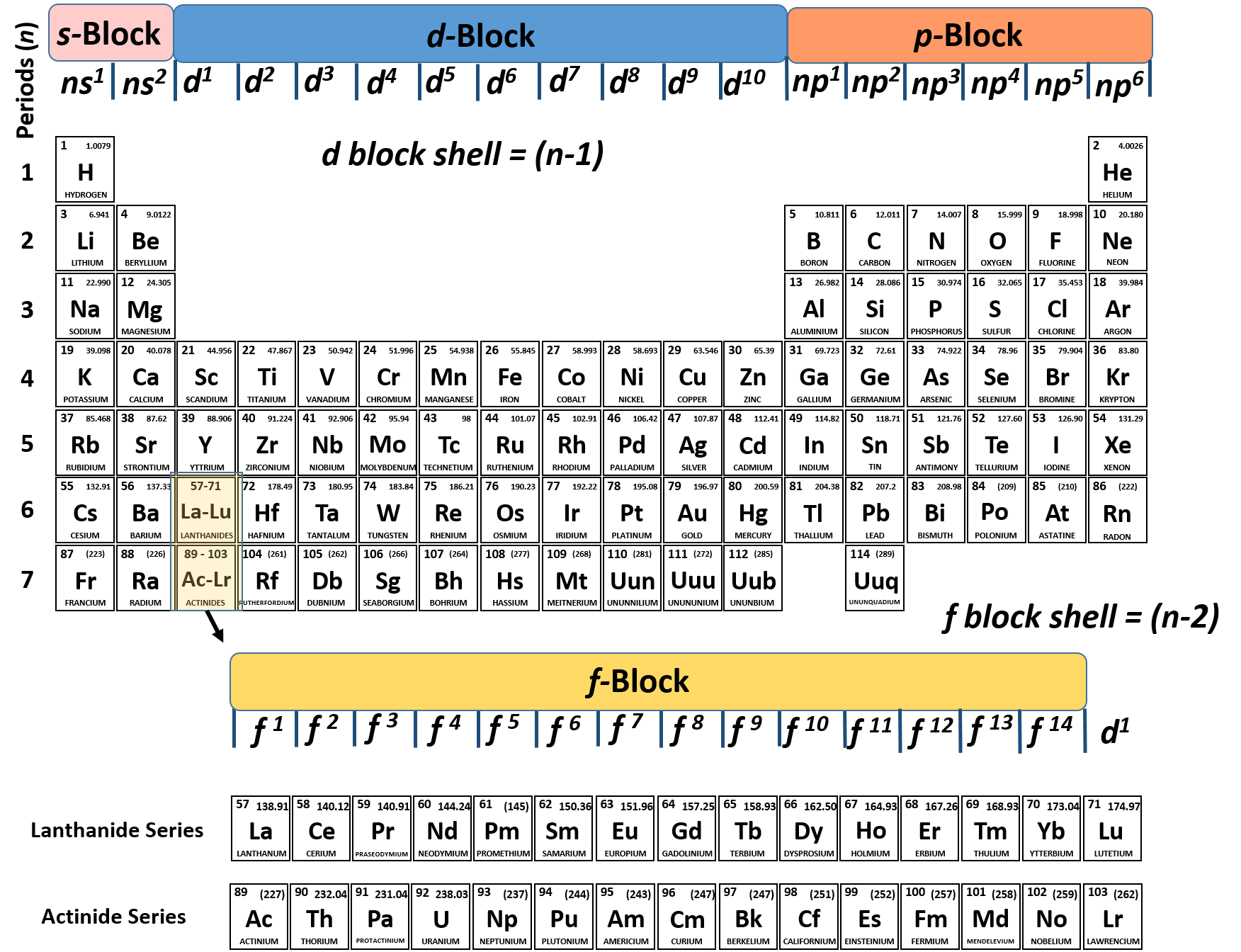
The shell diagram for a lithium atom is shown below. Because lithium’s final electron goes into the 2 s subshell, we write the electron configuration of a lithium atom as 1 s 22 s 1. The 2 s subshell holds a maximum of 2 electrons, and the 2 p subshell holds a maximum of 6 electrons. The second shell has two subshells, s and p, which fill with electrons in that order. Two of the lithium electrons can fit into the 1 s subshell, but the third electron must go into the second shell. The 1 s subshell cannot hold 3 electrons (because an s subshell can hold a maximum of 2 electrons), so the electron configuration for a lithium atom cannot be 1 s 3. Both electrons fit into the 1 s subshell because s subshells can hold up to 2 electrons therefore, the electron configuration for helium atoms is 1 s 2(spoken as “one-ess-two”). The electron configuration of a hydrogen atom is spoken out loud as “one-ess-one.” Electron configurations are shorthand descriptions of the arrangements of electrons in atoms. This structure is called an electron configuration. Thus, because a hydrogen atom has its single electron in the s subshell of the first shell, we use 1 s 1 to describe the electronic structure of hydrogen. We combine the shell and subshell labels when referring to the organization of electrons about a nucleus and use a superscript to indicate how many electrons are in a subshell. 
This first shell has only one subshell, which is labeled 1 s and can hold a maximum of 2 electrons. As shown in Table 2.6.1, the first shell, closest to the nucleus and with the lowest-energy electrons, is shell 1. We use numbers to indicate which shell an electron is in. It is the arrangement of electrons into shells and subshells that most concerns us here, so we will focus on that. Any s subshell can hold up to 2 electrons p, 6 d, 10 and f, 14. Different subshells hold a different maximum number of electrons.Thus, the first shell has only a single s subshell (called 1 s), the second shell has 2 s and 2 p subshells, the third shell has 3 s, 3 p, and 3 dand so forth. The subshells of each shell are labeled, in order, with the letters s, p, d, and f. The first shell has only one subshell, the second shell has two subshells, the third shell has three subshells, and so on. Shells are further divided into subsets of electrons called subshells.Shells do not have specific, fixed distances from the nucleus, but an electron in a higher-energy shell will spend more time farther from the nucleus than does an electron in a lower-energy shell. Generally the higher the energy of a shell, the farther it is (on average) from the nucleus. Electrons are organized according to their energies into sets called shells (labeled by the principle quantum number, n).We say that the energies of the electrons are quantized. Electrons in atoms can have only certain specific energies.It makes the following statements about electrons in atoms: The modern theory of electron behavior is called quantum mechanics. Do they move around the nucleus at random, or do they exist in some ordered arrangement? Describe how electrons are grouped within atoms.Īlthough we have discussed the general arrangement of subatomic particles in atoms, we have said little about how electrons occupy the space about the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
